acidPPcAcid phosphatase homologues |
|---|
| SMART accession number: | SM00014 |
|---|---|
| Description: | - |
| Interpro abstract (IPR000326): | This entry represents type 2 phosphatidic acid phosphatase (PAP2; EC 3.1.3.4 ) enzymes, such as phosphatidylglycerophosphatase B EC 3.1.3.27 from Escherichia coli. PAP2 enzymes have a core structure consisting of a 5-helical bundle, where the beginning of the third helix binds the cofactor [ (PUBMED:10835340) ]. PAP2 enzymes catalyse the dephosphorylation of phosphatidate, yielding diacylglycerol and inorganic phosphate [ (PUBMED:17079146) ]. In eukaryotic cells, PAP activity has a central role in the synthesis of phospholipids and triacylglycerol through its product diacylglycerol, and it also generates and/or degrades lipid-signalling molecules that are related to phosphatidate. Other related enzymes have a similar core structure, including haloperoxidases such as bromoperoxidase (contains one core bundle, but forms a dimer), chloroperoxidases (contains two core bundles arranged as in other family dimers), bacitracin transport permease from Bacillus licheniformis, glucose-6-phosphatase from rat. The vanadium-dependent haloperoxidases exclusively catalyse the oxidation of halides, and act as histidine phosphatases, using histidine for the nucleophilic attack in the first step of the reaction [ (PUBMED:12447906) ]. Amino acid residues involved in binding phosphate/vanadate are conserved between the two families, supporting a proposal that vanadium passes through a tetrahedral intermediate during the reaction mechanism. |
| Family alignment: |
There are 60557 acidPPc domains in 60341 proteins in SMART's nrdb database.
Click on the following links for more information.
- Evolution (species in which this domain is found)
- Disease (disease genes where sequence variants are found in this domain)
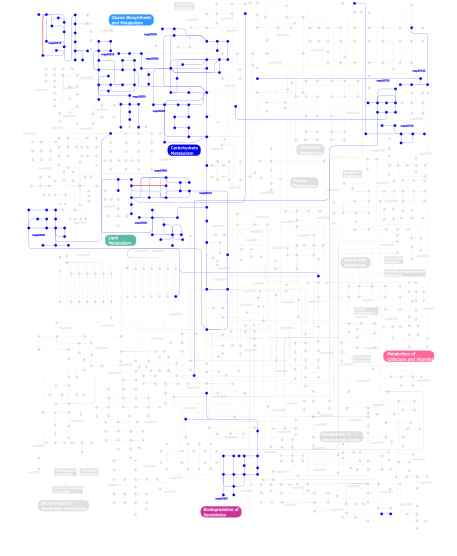
- Metabolism (metabolic pathways involving proteins which contain this domain)
- Structure (3D structures containing this domain)
- Links (links to other resources describing this domain)