POLBcDNA polymerase type-B family |
|---|
| SMART accession number: | SM00486 |
|---|---|
| Description: | DNA polymerase alpha, delta, epsilon and zeta chain (eukaryota), DNA polymerases in archaea, DNA polymerase II in e. coli, mitochondrial DNA polymerases and and virus DNA polymerases |
| Interpro abstract (IPR006172): | DNA is the biological information that instructs cells how to exist in an ordered fashion: accurate replication is thus one of the most important events in the life cycle of a cell. This function is performed by DNA- directed DNA-polymerases ( EC 2.7.7.7 ) by adding nucleotide triphosphate (dNTP) residues to the 5'-end of the growing chain of DNA, using a complementary DNA chain as a template. Small RNA molecules are generally used as primers for chain elongation, although terminal proteins may also be used for the de novo synthesis of a DNA chain. Even though there are 2 different methods of priming, these are mediated by 2 very similar polymerases classes, A and B, with similar methods of chain elongation. A number of DNA polymerases have been grouped under the designation of DNA polymerase family B. Six regions of similarity (numbered from I to VI) are found in all or a subset of the B family polymerases. The most conserved region (I) includes a conserved tetrapeptide with two aspartate residues. Its function is not yet known. However, it has been suggested [ (PUBMED:2461550) ] that it may be involved in binding a magnesium ion. All sequences in the B family contain a characteristic DTDS motif, and possess many functional domains, including a 5'-3' elongation domain, a 3'-5' exonuclease domain [ (PUBMED:8679562) ], a DNA binding domain, and binding domains for both dNTP's and pyrophosphate [ (PUBMED:9757117) ]. |
| GO function: | nucleotide binding (GO:0000166), DNA-directed DNA polymerase activity (GO:0003887), nucleic acid binding (GO:0003676) |
| Family alignment: |
There are 13646 POLBc domains in 13642 proteins in SMART's nrdb database.
Click on the following links for more information.
- Evolution (species in which this domain is found)
-
Taxonomic distribution of proteins containing POLBc domain.
This tree includes only several representative species. The complete taxonomic breakdown of all proteins with POLBc domain is also avaliable.
Click on the protein counts, or double click on taxonomic names to display all proteins containing POLBc domain in the selected taxonomic class.
- Cellular role (predicted cellular role)
-
Cellular role: replication
- Literature (relevant references for this domain)
-
Primary literature is listed below; Automatically-derived, secondary literature is also avaliable.
- Wang J, Yu P, Lin TC, Konigsberg WH, Steitz TA
- Crystal structures of an NH2-terminal fragment of T4 DNA polymerase and its complexes with single-stranded DNA and with divalent metal ions.
- Biochemistry. 1996; 35: 8110-9
- Display abstract
We report the crystal structure of an NH2-terminal 388-residue fragment of T4 DNA polymerase (protein N388) refined at 2.2 A resolution. This fragment contains both the 3'-5' exonuclease active site and part of the autologous mRNA binding site (J. D. Karam, personal communication). The structure of a complex between the apoprotein N388 and a substrate, p(dT)3, has been refined at 2.5 A resolution to a crystallographic R-factor of 18.7%. Two divalent metal ion cofactors, Zn(II) and Mn(II), have been located in crystals of protein N388 which had been soaked in solutions containing Zn(II), Mn(II), or both. The structure of the 3'-5' exonuclease domain of protein N388 closely resembles the corresponding region in the Klenow fragment despite minimal sequence identity. The side chains of four carboxylate residues that serve as ligands for the two metal ions required for catalysis are located in geometrically equivalent positions in both proteins with a rms deviation of 0.87 A. There are two main differences between the 3'-5' exonuclease active site regions of the two proteins: (I) the OH of Tyr-497 in the Klenow fragment interacts with the scissile phosphate in the active site whereas the OH of the equivalent tyrosine (Tyr-320) in protein N388 points away from the active center; (II) different residues form of the binding pocket for the 3'-terminal bases of the substrate. In the protein N388 complex the 3'-terminal base of p(dT)3 is rotated approximately 60 degrees relative to the position that the corresponding base occupies in the p(dT)3 complex with the Klenow fragment. Finally, a separate domain (residues 1-96) of protein N388 may be involved in mRNA binding that results in translational regulation of T4 DNA polymerase (Pavlov & Karam, 1994).
- Metabolism (metabolic pathways involving proteins which contain this domain)
-
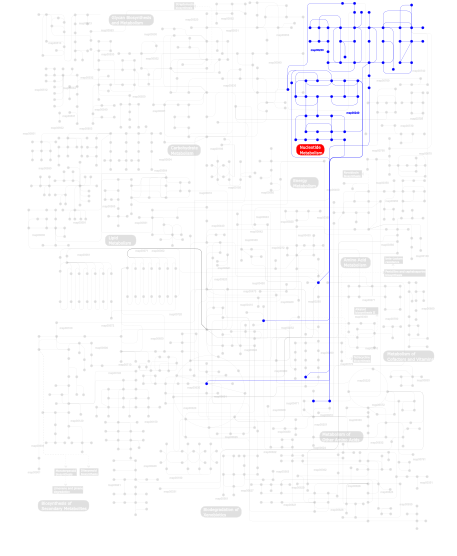
Click the image to view the interactive version of the map in iPath% proteins involved KEGG pathway ID Description 43.07 map03030 DNA replication 28.47  map00240
map00240Pyrimidine metabolism 28.47  map00230
map00230Purine metabolism This information is based on mapping of SMART genomic protein database to KEGG orthologous groups. Percentage points are related to the number of proteins with POLBc domain which could be assigned to a KEGG orthologous group, and not all proteins containing POLBc domain. Please note that proteins can be included in multiple pathways, ie. the numbers above will not always add up to 100%.
- Structure (3D structures containing this domain)
3D Structures of POLBc domains in PDB
PDB code Main view Title 1clq 
CRYSTAL STRUCTURE OF A REPLICATION FORK DNA POLYMERASE EDITING COMPLEX AT 2.7 A RESOLUTION 1d5a 
CRYSTAL STRUCTURE OF AN ARCHAEBACTERIAL DNA POLYMERASE D.TOK. DEPOSITION OF SECOND NATIVE STRUCTURE AT 2.4 ANGSTROM 1ig9 
Structure of the Replicating Complex of a Pol Alpha Family DNA Polymerase 1ih7 
High-Resolution Structure of Apo RB69 DNA Polymerase 1noy 
DNA POLYMERASE (E.C.2.7.7.7)/DNA COMPLEX 1noz 
T4 DNA POLYMERASE FRAGMENT (RESIDUES 1-388) AT 110K 1q8i 
Crystal structure of ESCHERICHIA coli DNA Polymerase II 1q9x 
Crystal structure of Enterobacteria phage RB69 gp43 DNA polymerase complexed with tetrahydrofuran containing DNA 1q9y 
CRYSTAL STRUCTURE OF ENTEROBACTERIA PHAGE RB69 GP43 DNA POLYMERASE COMPLEXED WITH 8-OXOGUANOSINE CONTAINING DNA 1qht 
DNA POLYMERASE FROM THERMOCOCCUS SP. 9ON-7 ARCHAEON 1qqc 
CRYSTAL STRUCTURE OF AN ARCHAEBACTERIAL DNA POLYMERASE D.TOK 1s5j 
Insight in DNA Replication: The crystal structure of DNA Polymerase B1 from the archaeon Sulfolobus solfataricus 1tgo 
THERMOSTABLE B TYPE DNA POLYMERASE FROM THERMOCOCCUS GORGONARIUS 1waf 
DNA POLYMERASE FROM BACTERIOPHAGE RB69 1waj 
DNA POLYMERASE FROM BACTERIOPHAGE RB69 1wn7 
Crystal structure of archaeal family B DNA polymerase mutant 1wns 
Crystal structure of family B DNA polymerase from hyperthermophilic archaeon pyrococcus kodakaraensis KOD1 1xhx 
Phi29 DNA Polymerase, orthorhombic crystal form 1xhz 
Phi29 DNA polymerase, orthorhombic crystal form, ssDNA complex 1xi1 
Phi29 DNA polymerase ssDNA complex, monoclinic crystal form 2atq 
RB69 single-stranded DNA binding protein-DNA polymerase fusion 2dtu 
Crystal structure of the beta hairpin loop deletion variant of RB69 gp43 in complex with DNA containing an abasic site analog 2dy4 
Crystal structure of RB69 GP43 in complex with DNA containing Thymine Glycol 2ex3 
Bacteriophage phi29 DNA polymerase bound to terminal protein 2gv9 
Crystal structure of the Herpes Simplex virus type 1 DNA polymerase 2jgu 
crystal structure of DNA-directed DNA polymerase 2oyq 
Crystal structure of RB69 gp43 in complex with DNA with 5-NIMP opposite an abasic site analog 2ozm 
Crystal structure of RB69 gp43 in complex with DNA with 5-NITP opposite an abasic site analog 2ozs 
Crystal structure of RB69 gp43 in complex with DNA with dATP opposite dTMP 2p5g 
Crystal structure of RB69 gp43 in complex with DNA with dAMP opposite an abasic site analog in a 21mer template 2p5o 
Crystal structure of RB69 GP43 in complex with DNA containing an abasic site analog 2py5 
Phi29 DNA polymerase complexed with single-stranded DNA 2pyj 
Phi29 DNA polymerase complexed with primer-template DNA and incoming nucleotide substrates (ternary complex) 2pyl 
Phi29 DNA polymerase complexed with primer-template DNA and incoming nucleotide substrates (ternary complex) 2pzs 
Phi29 DNA polymerase complexed with primer-template DNA (post-translocation binary complex) 2vwj 
Uracil Recognition in Archaeal DNA Polymerases Captured by X-ray Crystallography. 2vwk 
Uracil Recognition in Archaeal DNA Polymerases Captured by X-ray Crystallography. V93Q polymerase variant 2xhb 
Crystal structure of DNA polymerase from Thermococcus gorgonarius in complex with hypoxanthine-containing DNA 3a2f 
Crystal Structure of Pyrococcus furiosus DNA polymerase/PCNA monomer mutant complex 3cfo 
Triple Mutant APO structure 3cfp 
Structure of the replicating complex of a POL Alpha family DNA Polymerase, ternary complex 1 3cfr 
Structure of the replicating complex of a POL Alpha family DNA Polymerase, ternary complex 2 3cq8 
Ternary complex of the L415F mutant RB69 exo(-)polymerase 3iay 
Ternary complex of DNA polymerase delta 3k57 
Crystal structure of E.coli Pol II-normal DNA-dATP ternary complex 3k58 
Crystal structure of E.coli Pol II-normal DNA-dTTP ternary complex 3k59 
Crystal structure of E.coli Pol II-normal DNA-dCTP ternary complex 3k5l 
Crystal structure of E.coli Pol II-abasic DNA-dATP Lt(0, 3) ternary complex 3k5m 
Crystal structure of E.coli Pol II-abasic DNA-ddGTP Lt(-2, 2) ternary complex 3k5n 
Crystal structure of E.coli Pol II-abasic DNA binary complex 3k5o 
Crystal structure of E.coli Pol II 3kd1 
Closed binary complex of an RB69 gp43 fingers domain mutant complexed with an acyclic GMP terminated primer template pair. 3kd5 
Closed ternary complex of an RB69 gp43 fingers domain mutant complexed with an acyclic GMP terminated primer template pair and phosphonoformic acid. 3l8b 
Crystal structure of a replicative DNA polymerase bound to the oxidized guanine lesion guanidinohydantoin 3lds 
Crystal structure of RB69 gp43 with DNA and dATP opposite 8-oxoG 3lzi 
RB69 DNA Polymerase (Y567A) ternary complex with dATP Opposite 7,8-dihydro-8-oxoguanine 3lzj 
RB69 DNA Polymerase (Y567A) ternary complex with dCTP Opposite 7,8-Dihydro-8-oxoguanine 3maq 
Crystal structure of E.coli Pol II-normal DNA-dGTP ternary complex 3nae 
RB69 DNA Polymerase (Y567A) Ternary Complex with dATP Opposite Guanidinohydantoin 3nci 
RB69 DNA Polymerase Ternary Complex with dCTP Opposite dG at 1.8 angstrom resolution 3ndk 
RB69 DNA Polymerase (Y567A) Ternary Complex with dCTP Opposite dG 3ne6 
RB69 DNA Polymerase (S565G/Y567A) Ternary Complex with dCTP Opposite dG 3ngi 
RB69 DNA Polymerase (Y567A) Ternary Complex with dTTP Opposite dG 3nhg 
RB69 DNA Polymerase (S565G/Y567A) Ternary Complex with dTTP Opposite dG 3qei 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dCTP Opposite Difluorotoluene Nucleoside 3qep 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dTTP Opposite Difluorotoluene Nucleoside 3qer 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dATP Opposite Difluorotoluene Nucleoside 3qes 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dGTP Opposite Difluorotoluene Nucleoside 3qet 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dTTP Opposite dT 3qev 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dCTP Opposite dT 3qew 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dDTP Opposite dT 3qex 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dGTP Opposite dT 3qnn 
RB69 DNA Polymerase (Y567A) Ternary Complex with dGT Opposite 3tCo 3qno 
RB69 DNA Polymerase (Y567A) Ternary Complex with dATP Opposite 3tCo 3rma 
Crystal Structure of a replicative DNA polymerase bound to DNA containing Thymine Glycol 3rmb 
Crystal Structure of a replicative DNA polymerase bound to DNA containing Thymine Glycol 3rmc 
Crystal Structure of a replicative DNA polymerase bound to DNA containing Thymine Glycol 3rmd 
Crystal Structure of a replicative DNA polymerase bound to DNA containing Thymine Glycol 3rwu 
RB69 DNA Polymerase (Y567A) Ternary Complex with dATP Opposite Difluorotoluene Nucleoside 3s9h 
RB69 DNA Polymerase Triple Mutant(L561A/S565G/Y567A) ternary complex with dUpNpp and a dideoxy-terminated primer in the presence of Ca2+ 3scx 
RB69 DNA Polymerase Triple Mutant(L561A/S565G/Y567A) Ternary Complex with dUpNpp and a Deoxy-terminated Primer in the Presence of Ca2+ 3si6 
RB69 DNA Polymerase Triple Mutant (L561A/S565G/Y567A) Ternary Complex with dUpNpp and a Deoxy-terminated Primer in the presence of Mg2+ 3sjj 
RB69 DNA Polymerase Triple Mutant (L561A/S565G/Y567A) Ternary Complex with dUpNpp and a Deoxy-terminated Primer in the presence of Mn2+ 3snn 
RB69 DNA Polymerase (L561A/S565G/Y567A) Ternary Complex with dCTP Opposite dG in the presence of Mg2+ 3spy 
RB69 DNA Polymerase(L415A/L561A/S565G/Y567A) Ternary Complex with dUpCpp Opposite dA 3spz 
DNA Polymerase(L415A/L561A/S565G/Y567A) Ternary Complex with dUpCpp Opposite dA (Ca2+) 3sq0 
DNA Polymerase(L561A/S565G/Y567A) Ternary Complex with dUpNpp Opposite dA (Mn2+) 3sq1 
RB69 DNA Polymerase Ternary Complex with dUpCpp Opposite dA 3sq2 
RB69 DNA Polymerase Ternary Complex with dTTP Opposite 2AP (AT rich sequence) 3sq4 
RB69 DNA Polymerase Ternary Complex with dTTP Opposite 2AP (GC rich sequence) 3sun 
RB69 DNA Polymerase (Y567A) Ternary Complex with dTTP Opposite 2AP (AT rich sequence) 3suo 
RB69 DNA Polymerase (Y567A) Ternary Complex with dTTP Opposite 2AP (GC rich sequence) 3sup 
RB69 DNA Polymerase (Y567A) Ternary Complex with dCTP Opposite 2AP (GC rich sequence) 3suq 
RB69 DNA Polymerase (Y567A) Ternary Complex with dCTP Opposite 2AP (AT rich sequence) 3tab 
5-hydroxycytosine paired with dGMP in RB69 gp43 3tae 
5-hydroxycytosine paired with dAMP in RB69 gp43 3taf 
5-fluorocytosine paired with ddGMP in RB69 gp43 3tag 
5-fluorocytosine paired with dAMP in RB69 gp43 3uiq 
RB69 DNA Polymerase Ternary Complex containing dUpNpp 4ahc 
Crystal Structure of an Evolved Replicating DNA Polymerase 4ail 
Crystal Structure of an Evolved Replicating DNA Polymerase 4b08 
Yeast DNA polymerase alpha, Selenomethionine protein 4dtj 
RB69 DNA Polymerase Ternary Complex with dTTP Opposite an Abasic Site and ddT/dA as the Penultimate Base-pair 4dtm 
RB69 DNA Polymerase Ternary Complex with dCTP Opposite an Abasic Site and ddG/dC as the Penultimate Base-pair 4dtn 
RB69 DNA Polymerase Ternary Complex with dATP Opposite an Abasic Site and ddA/dT as the Penultimate Base-pair 4dto 
RB69 DNA Polymerase Ternary Complex with dCTP Opposite an Abasic Site and ddA/dT as the Penultimate Base-pair 4dtp 
RB69 DNA Polymerase Ternary Complex with dGTP Opposite an Abasic Site and ddA/dT as the Penultimate Base-pair 4dtr 
RB69 DNA Polymerase Ternary Complex with dATP Opposite an Abasic Site and ddC/dG as the Penultimate Base-pair 4dts 
RB69 DNA Polymerase Ternary Complex with dCTP Opposite an Abasic Site and ddC/dG as the Penultimate Base-pair 4dtu 
RB69 DNA Polymerase Ternary Complex with dGTP Opposite an Abasic Site and ddC/dG as the Penultimate Base-pair 4dtx 
RB69 DNA Polymerase Ternary Complex with dTTP Opposite an Abasic Site and ddC/dG as the Penultimate Base-pair 4du1 
RB69 DNA Polymerase Ternary Complex with dATP Opposite dT 4du3 
RB69 DNA Polymerase Ternary Complex with dDTP Opposite dT with 3-Deaza-adenine at the N-1 Position of Template Strand 4du4 
RB69 DNA Polymerase Ternary Complex with dATP Opposite dT with 3-Deaza-adenine at the N-3 Position of Primer Strand 4e3s 
RB69 DNA Polymerase Ternary Complex with dQTP Opposite dT 4fj5 
RB69 DNA polymerase ternary complex with dATP/dT 4fj7 
RB69 DNA polymerase ternary complex with dGTP/dT 4fj8 
RB69 DNA polymerase ternary complex with dCTP/dT 4fj9 
RB69 DNA polymerase ternary complex with dTTP/dT 4fjg 
RB69 DNA polymerase ternary complex with dATP/dC 4fjh 
RB69 DNA polymerase ternary complex with dGTP/dC 4fji 
RB69 DNA polymerase ternary complex with dcTP/dC 4fjj 
RB69 DNA polymerase ternary complex with dTTP/dC 4fjk 
RB69 DNA polymerase ternary complex with dATP/dA 4fjl 
RB69 DNA polymerase ternary complex with dGTP/dA 4fjm 
RB69 DNA polymerase ternary complex with dCTP/dA 4fjn 
RB69 DNA polymerase ternary complex with dTTP/dA 4fjx 
RB69 DNA polymerase ternary complex with dATP/dG 4fk0 
RB69 DNA polymerase ternary complex with dCTP/dG 4fk2 
RB69 DNA polymerase ternary complex with dTTP/dG 4fk4 
RB69 DNA polymerase ternary complex with dGTP/dG 4flt 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4flu 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4flv 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4flw 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4flx 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4fly 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4flz 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4fm0 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4fm1 
Pyrococcus abyssi B family DNA polymerase bound to a dsDNA, in edition mode 4fm2 
Pyrococcus abyssi B family DNA polymerase (triple mutant) bound to a dsDNA, in edition mode 4fvm 
Crystal structure of yeast DNA polymerase alpha 4fxd 
Crystal structure of yeast DNA polymerase alpha bound to DNA/RNA 4fyd 
Crystal structure of yeast DNA polymerase alpha bound to DNA/RNA and dGTP 4i9l 
Crystal structure of the D714A mutant of RB69 DNA polymerase 4i9q 
Crystal structure of the ternary complex of the D714A mutant of RB69 DNA polymerase 4j2a 
RB69 DNA Polymerase L415A Ternary Complex 4j2b 
RB69 DNA Polymerase L415G Ternary Complex 4j2d 
RB69 DNA Polymerase L415K Ternary Complex 4j2e 
RB69 DNA Polymerase L415M Ternary Complex 4k8x 
Binary complex of 9N DNA polymerase in the replicative state 4k8z 
KOD Polymerase in binary complex with dsDNA 4khn 
Crystal structure of the ternary complex of the D714A mutant of RB69 DNA polymerase 4khq 
Ternary complex of RB69 mutant L415F wit DUMPNPP 4khs 
Ternary complex of RB69 mutant L415F with a ribonucleotide at 0 position 4khu 
Ternary complex of rb69 mutant L415F with a ribonucleotide at -1 position 4khw 
Ternary complex of RB69 mutant L415F with ribonucleotide at -2 position 4khy 
Ternary complex of rb69 mutant L415F with ribonucleotide at -3 position 4ki4 
Ternary complex of rb69 mutant L415F with ribonucleotides at 0 and -1 position 4ki6 
Ternary complex of rb69 mutant l415f with ribonucleotides at -1 and -2 position 4m3r 
RB69 DNA polymerase ternary complex with dT/dG at position n-1 of primer/template duplex 4m3t 
RB69 DNA polymerase ternary complex with dT/dG at position n-2 of primer/template duplex 4m3u 
RB69 DNA polymerase ternary complex with dT/dG at position n-3 of primer/template duplex 4m3w 
RB69 DNA polymerase ternary complex with dT/dG at position n-4 of primer/template duplex 4m3x 
RB69 DNA polymerase ternary complex with dT/dG at position n-5 of primer/template duplex 4m3y 
RB69 DNA polymerase ternary complex with dG/dT at position n-1 of primer/template duplex 4m3z 
RB69 DNA polymerase ternary complex with dG/dT at position n-2 of primer/tempLate duplex 4m41 
RB69 DNA polymerase ternary complex with dG/dT at position n-3 of primer/tempLate duplex 4m42 
RB69 DNA polymerase ternary complex with dG/dT at position n-4 of primer/tempLate duplex 4m45 
RB69 DNA polymerase ternary complex with dG/dT at position n-5 of primer/template duplex 4m8o 
TERNARY COMPLEX OF DNA POLYMERASE EPSILON WITH AN INCOMING dATP 4ptf 
Ternary crystal structure of yeast DNA polymerase epsilon with template G 4q5v 
4Q5V 4qcl 
4QCL 5exr 
5EXR 5iud 
5IUD - Links (links to other resources describing this domain)
-
INTERPRO IPR006172 PFAM DNA_pol_B PROSITE DNA_POLYMERASE_B

